Product Name: Cupric carbonate basic
Synonym: Basic copper carbonate; Copper (II) carbonate basic; Copper (II) hydroxide carbonate
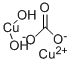
Molecular Structure:
Specifications:
| Item | Specifications |
| CAS No. | 12069-69-1 |
| Molecular Formula | CO3.Cu.CuH2O2 |
| Molecular Weight | 221.11 |
| Melting point | 200 °C |
| Density | 4 |
| Form | Solid |
| Color | green |
| Specific gravity | 4 |
| Water solubility | Insoluble |
Packing: 200 kg/Drum
Function:
Alkaline copper carbonate, also known as copper carbonate, is peacock green in color, hence the name malachite, and is a valuable mineral gemstone. It is a substance produced by the reaction of copper with substances such as oxygen, carbon dioxide and water in the air, also known as patina, and is bright green in color. When heated in air, it decomposes into copper oxide, water and carbon dioxide. It dissolves in acids and produces the corresponding copper salts. Also dissolves in cyanide, ammonium salts and alkali metal carbonate aqueous solutions to form copper complexes. When boiled in water or heated in strong alkali solution, it can produce brown copper oxide, which decomposes into black copper oxide at 200°C. Unstable in the hydrogen sulfide atmosphere, can react with hydrogen sulfide to produce copper sulfide. Alkaline copper carbonate exists in more than a dozen forms of compounds according to the ratio of CuCO3:H2O. It exists in nature in the form of malachite.
Application:
Used in pyrotechnics, pesticides, pigments, feed, fungicides, antiseptics and other industries and the manufacture of copper compounds
Used as analytical reagent and insecticide
Used in industries such as catalysts, pyrotechnics, pesticides, pigments, feeds, fungicides, electroplating, anticorrosion, and the manufacture of copper compounds
Storage:
Stored in a cool dry and ventilated place







Reviews
There are no reviews yet.